Control and monitoring of plants of eutrophication
May 7, 2009
Both systems, especially plants of absorption, can be designed to produce a content of oxygen much more low, although it is not economical. Plants of eutrophication ('of-oxo') can alternatively be used to remove the remains of oxygen from the products of these plants.
The plants of eutrophication function by adding hydrogen to nitrogen of low grade, by reacting the resulting mixture to pass through the column of catalytically active material. Hydrogen reacts with the residual oxygen to form water. The following chemical reaction describes the reaction:
2 H 2 + O2 + N2 = 2H2O + N2
If necessary, the gas can impersonate a dryer. For many technical reasons and security, many processes using the gas produced by this system do not tolerate any excess hydrogen in the product. The problem, therefore, is to add hydrogen to react with oxygen.
The most common approaches and the solution raised by Hitech
One of the most common problem approaches has been monitoring the concentration of oxygen in the product of the generator of nitrogen and its flow to the reactor of eutrophication. A microprocessor look these values and extract the ratio of the hydrogen required to react with the oxygen flow. The required flow is then adjusted through a mass flow meter in the line of hydrogen. As a security check, products monitored through an Analyzer of hydrogen which is at best +/-50 ppm.
The method works, but has 2 big drawbacks: 1) is expensive, requiring analyzers rotámetro, computers, precision and flow-meters gap; (and 2) the result is that the product contains relatively large quantities of hydrogen.
An alternative approach is to use a Hitech Instruments oxygen Analyzer with zirconium sensor. Cell of zirconia oxygen analyzers have a number of unique attributes, two of which make it ideal for this application. First of all, they can measure oxygen at concentrations from 1 to 10-23 atmospheres, and secondly, they can do this with a response time of fractions of seconds. They measure the concentration of oxygen in the same way that pH electrodes measure the acidity.
And in the same way that pH electrode is used to monitor an acid neutralization by alkali, may used a zirconia sensor to monitor the "neutralization" of oxygen with hydrogen. The point of "neutralization" occurs when the items are exactly in the correct proportion to not produce any excess. This point is usually called the stoichiometric point. In it, the concentrations of oxygen and hydrogen are due entirely to the dissociation of water produced by the reaction and are of the order of parts per trillion.
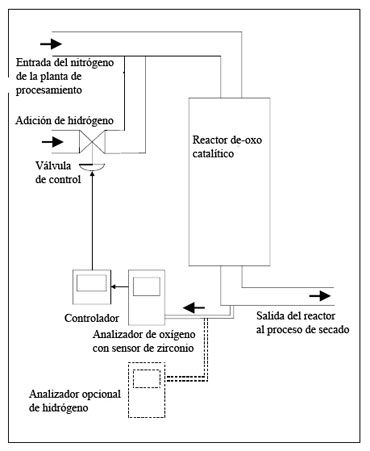
Figure 1 shows the general system for monitoring and control of-oxo plant using an Analyzer of zirconium from Hitech Instruments. Using this approach, you get a lower cost with a much simpler system controller. If required, additional hydrogen analyser can be incorporated to monitor a situation of failure, for example, in the case that fails the reactor of-oxo.









