Degradation and conservation of vidrieras mediaeval
31 May 2011
Before establishing the criteria for intervention and conservation it is necessary to determine the pathologies of the stained-glass window according to the following points:
- Observation of the alterations of the glass
- Analysis of the composition of the glass and its alterations
- Characterization and identification of defects
- Determination of its causes
- Choice of cleaning and restoration treatments
- Procedures for the protection
- Maintenance and periodic inspection
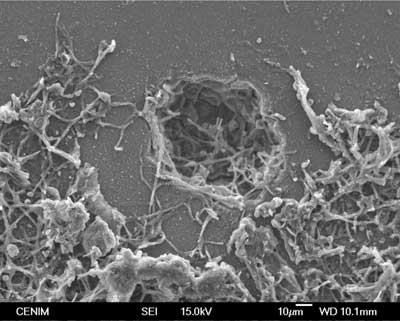
The observation of the damage and pathologies of the stained-glass materials will make it possible to carry out a prior assessment. To this end are used various characterization techniques that are not destructive or microdestructivas, to the extent possible. The State of conservation is inspected first by observation with a binocular magnifying glass and then a regular optical microscope (MO). A scanning electron microscope (SEM) scan that can be studied surface flaws (cracks, punctures, craters, spots), and the deposits formed as a result of the weathering and/or chemical degradation is used if necessary. This observation should be accompanied by a chemical analysis of the inside of the glass and its surface where corrosion processes have been initiated. This makes it possible to establish the composition differences between the interior and the surface of the glass, and assess the degree of alteration suffered. The depth of the attack and the selective extraction of certain components of the glass can be studied by spectrometry of scattering of x-ray X (EDS), technical energy which is usually coupled to a SEM, or secondary ions (SIMS) mass spectrometry.

Causes of deterioration
The causes of alteration and degradation of the glass may be characteristic of the material (intrinsic) or external to it (extrinsic).
Intrinsic causes
- Type of glass
- Time and place of manufacture
- Mechanism and intensity of chemical attack
- Nature of the deposits formed
- Cleaning and protection treatments to be used
- Any reproduction of parts for replacement

The major components of the medieval glass are the oxide of Silicon SiO2, CaO calcium oxide and oxide of potassium (K)2or, in other words are calcium potassium silicate glass. Other components of these glasses are the oxide of phosphorus P2or5 and PbO lead oxide. In the medieval glass the content of oxide from sodium Na2O is very low (0 - 1% by weight). It has shown that only when the sum of the percentages of forming oxides glass (oxides of silicon, aluminum and phosphorus) is greater than 60% by weight, the glasses are preserved correctly. Below this value the glasses are attacked by weathering and pollution, and on its surface appear bites, craters, crusts of corrosion, deposits, etc. On the other hand, the proportion and nature of alkali oxides in the glass are very important, because above 20% by weight, they give rise to no stable glass; and, as regards the type of alkaline oxide, potassium glass (such as the medieval) are much less resistant than the soda glass (for example, the Roman glass or modern).

Extrinsic causes
These causes act in the most vulnerable points of the glass and accelerate their attack. They can be chemical, physical and biological causes.
Chemical causes
Chemical degradation of the glass is the result of the progressive weathering, especially in the outer side of the stained glass Windows, long a very prolonged and aggravated in the last century, due to the increase in air pollution. The corrosion of glass begins on its surface, preferably in areas with microfisuras or mechanical injury attributable to defects or tensions, by contact with the humidity, even amidst a neutral pH. The surface absorbs water and is hydrated to form a layer of silica gel in which dissolve the gaseous pollutants in the air. These gases (CO2, OSx, notx, etc.), in combination with the humidity, they acidic environment next to the glass so that the ions H of these acids absorbed by the glass at the expense of the removal of alkali ions of the glassy network (process of ion-exchange, in the acid attack). As a result of this process the surface of the glass is desalcalinizada and the next glass environment is enriched in alkaline substances that will first neutralizing and increasing pH then to core values. At high pH attack is very dangerous, because that destroy joints Si-O - are those that form the glass network (attack in the Basic or alkaline). This mechanism of attack explains why the medieval calcium potassium silicate glass have attacked more than the glass of calcium sodium silicate from other eras. The reason is that the ions potassium K+ extracted far more easily than the ions sodium Na+ and accelerate the destruction of the glassy network.

The first product of the corrosion of glass are the alkali hydroxides which subsequently become carbonates, due to the CO2 environmental, and finally form sulphates by interaction with the pollution from sulphur oxides. Soluble salts deposits are removed with the rain, while insoluble products remain forming crystalline deposits. The corrosion of glass is manifested by the appearance of isolated bites that grow and become increasingly deep, numerous and interconnected craters. In these craters are deposited the corrosion products which end up forming a more or less uniform layer or a layer of porous corrosion which does not prevent that degradation continue in depth. The consequences are a reduction of the original thickness of the glass, the loss of its transparency and masking of the colors. The most common compounds that are formed in the crusts of corrosion are hydrated silica (silica gel), sulfates of calcium (plaster, singenita) and calcium carbonate. Calcium carbonate can also come from the fillers used for sealing between the fragments of glass and the network of emplomado, and in a subsequent process can lead to calcium oxalate by reaction with oxalic acid generated by certain microorganisms. In addition to these corrosion products of the own glass, there are other compounds from the oxidation of bars of subject, the frames and the network of emplomado, which are washed out by rain and deposited, and integrated into the crusts of corrosion.
There is a well-defined boundary between the corrosion layer and the underlying glass, they both are linked by a continuous intercara glass attacked unevenly. By that when he carried out a cleaning must proceed carefully and always avoid the complete elimination of the first layer of silica gel which, in reality, protects the glass of a subsequent attack.
The inner side of the stained glass windows often present a more acceptable than the exterior condition, since it has been protected from pollutants. Without however, it is not exempt from proper alterations, in this case, to the condensation favored by human concentrations, the differences in temperature between the inside and outside, etc. Apparently harmless condensation droplets remain long periods deposited on the surface of the glass where they produce the extraction of alkali ions, so the chemical attack triggers as indicated above. The most sensitive elements of the stained glass Windows are the pictorial layers and decorations based on grisailles (vitrificables paintings). The grisailles are generally applied by the inner side of the stained glass Windows and its chemical composition is very different from the of the medieval glass. They are usually formed by mixtures of oxides and easily vitrificables salts containing dyes of dark tones. Applied on the surface of the glass in the form of suspensions and were baked at 500-600 ° C. The usual fluxes of the grisailles are oxides of lead and iron, increased chemical resistance than the alkali fluxes of the glass base, so the stability of the grisailles is often greater than the underlying glass that, ultimately, protect it from corrosion.

Physical causes
The main physical causes affecting the conservation of the medieval stained glass Windows are mechanical damage and optical causes. Glass as a fragile material fracture easily by impacts, acts of vandalism, vibration, pressure of the wind, internal tensions and tensions of the subject-metallic elements. The mechanical resistance of the glass is reduced by the existence of small cracks and the interaction with the atmosphere. The microfisuras originate on the surface by small injuries due to abrasion, local thermal stress, differences in contraction in areas with small differences in composition, etc. They can be seen at the electron microscope and act as multipliers centres of tension on the surface of the glass. Any mechanical effort is concentrated at the apex of the crack where it starts and propagates the rupture. A fissure can grow for a long time until it reaches a critical size for which the catastrophic rupture occurs. The atmospheric moisture is an agent that promotes the spread of the cracks, as it degrades the vertex of the fissure chemically destroying the glass network. The three main factors that determine this progressive mechanical destruction of the glassy network are the intensity of the applied load, the concentration of environmental water and time of performance of moisture.
Optical causes must be cited progressive dimming, which manifests itself especially in glass containing manganese oxide. This oxide is found in many medieval glass as impurity provided by raw materials providing the alkali flux, but also the Mn2or3 was used intentionally as colorant to produce the color of the pink flesh. Mn ions3+ can be drawn from the glass towards the surface where is fotooxidan at ions Mn4 + effect of sunlight. Insoluble2 that gives dark brown build up in layers of corrosion as MnO. This phenomenon, which may occur in a few years, is known as enmarronamiento.

Biological causes
Glass biogenic crusts are formed by organisms and microorganisms whose metabolites involved in various reactions with the components of the glass that serves as their support. The biodeterioration of the internal faces is very different from the external faces of the stained glass Windows. Occur in the internal bites due to the condensation where the microorganisms are developed, and can be of several millimeters in diameter and be filled with salts, hydroxides, bacteria, dust, etc. The biodeterioration is particularly serious when it occurs on surfaces decorated with pictorial layers containing organic type binders, as the organic matter favours the development of micro-organisms. On the external sides are algae, lichens and mosses that combined with high moisture attack the glass and retain water which intensifies the chemical deterioration. On the other hand, micro-organisms also produce a chemical deterioration due to their own metabolism: organic acids, inorganic acids and chelate that give rise to a change in the pH of the surface of the glass and the production of pigments that alter the aesthetics of the stained-glass window. The main bodies that have an impact on the glass of the stained glass Windows are: sulphurous bacteria, nitrifying bacteria, ferrobacterias, fungi, algae, lichens and mosses.
Cleaning and restoration
In the restoration of the stained glass Windows should take into account that attacked layers of glass may not recover and that, therefore, is limited to a clean and respectful intervention. These tasks are multidisciplinary and must be carried out in close collaboration of art historians, architects, artists, conservators and scientists specializing in glass. Successive operations are as follows: cleaning (mechanical removal of dirt, reactions of dissolution, decalcification, etc.), sealing of cracks and edges, Union of broken fragments, reengrosado adelgazados glass, establishment and consolidation of grisailles, General design, renovation of the emplomado (if necessary), elimination of leads of breakage and replacement (if necessary) of the metal fastening elements.
It is important to repeat that for the Elimination of layers of alteration should be avoided that destroy or alter the protective film of silica gel, which is the first layer attached to the unchanged surface of the glass. The preservation of this layer will protect the glass of subsequent chemical attacks. After the mechanical cleaning with compressed air and brushes and soft brushes, is carried out in the wet cleaning using cloth impregnated with a mixture to 50% by volume of ethyl alcohol and water. Cleaning laser with good results has also been used in the last decade. The dissolution of calcareous crusts can be provided with chemical calcium complejantes, as the AEDT, phosphates, tiosulfatos, bicarbonates, etc., although its use should be limited to extreme cases, because many specialists question its use.
Procedures for the protection
Procedures for the protection of the stained glass Windows are even more questionable than the restoration. The application of coatings or any active treatment on the surface of the glass has been highly controversial. However, its use is recommended when there are cracks whose spread is necessary to stop. In this case applied water-proof sealants, where previously it has ensured the lack of humidity. In any case, it is necessary to perform tests with model glass prepared with the same chemical composition as the original glass. Protective coatings must meet the following conditions: good adhesion, good barrier against water and pollutants, reversibility, optical neutrality and stability to the solar radiation, moisture and contaminants. Use of polymeric, sealants, consolidantes materials, etc. must be made when relevant prior trials have been carried out in order to verify compliance with the above listed conditions. The most widely used coatings are based on resins Acrylic polymers, silicone, epoxy, layers prepared by the procedure sol-gel (alkoxides of silicon or titanium or zirconium), ormocers (orgánico-inorgánicos hybrid materials derived from silicates, Silanes, etc.).

Currently system employed and innocuous for the protection of the stained glass is the placement of a colourless glazing external instead of stained-glass window, while it is retracted into the interior of the building. Thus the stained glass is isolated from the atmosphere, atmospheric phenomena and pollution, so its weathering is practically stopped. Between the window and the exterior glazing is necessary to let a camera of ventilation of at least some 15 centimetres to facilitate natural ventilation to prevent the condensation.
In addition to the controls of temperature and relative humidity, it is advisable to carry out an evaluation with sensors that can detect the presence of certain pollutant gases, or sensors that tell of the generation of a harmful environment for risk of acid shock. In this case the recently developed and patented sensors of environmental acidity in the history Institute of the CSIC are very useful. These sensors include a film dopada with a dye sensitive to changes in overall acidity of the atmosphere. His answer is optical and produces a color change that can be evaluated qualitatively using a preset color scale, or by using the corresponding curves of calibration performed with the data of visible absorption Spectra recorded at different values of environmental acidity.

Dr. Maria Angeles Villegas, degree in inorganic chemistry, is scientific titular of the Consejo Superior de Investigaciones Científicas (CSIC) since 1990. She was 17 years old intended in the Institute of ceramics and glass, and 7 years in the National Centre for metallurgical research. From the month of July 2007, he develops his research work in Institute history, specifically the Centre for human and social sciences, a multidisciplinary team that integrates chemists, engineers and archaeologists, which focuses on the preservation and conservation of our heritage.
Thus, his team has studied stained glass of the Cathedral of León, Vitoria and Sevilla of the Carthusian monastery of Miraflores and the monastery of San Juan of the Reyes de Toledo, among other buildings, as well as the behavior and State of metals from the train station of Aranjuez and of Atocha. "Materials suffer serious problems of degradation and, even, corrosion, in the case of metals." "Therefore, is very important to establish the physicochemical that corrosion mechanisms because if we know them, we know how to stop its deterioration," explains Dr.










































